|
When the pH of the solution of a weak acid is equal to the pK' of the acid, then 50% of the acid is in the undissociated form, HA, and 50% is in the dissociated form, H + + A. This equation can be used to calculate the pH of everything from water, to the blood in our bodies, and is essential to biochemistry.ĭescribes the relationship between pH and the extent of dissociation of a weak acid. Untuk dapat menggunakan persamaan Henderson - Hasselbalch, yang untuk penampal yang mengandungi asas lemah dan konjugasinya kelihatan seperti. It is also used to find the isoelectric point of proteins, as well as the equilibrium pH in acid-base reactions. Anda berurusan dengan penyelesaian penampan yang mengandungi piridin, 'C' 5 'H' 5 'N', asas yang lemah, dan pyridinium chloride, 'C' 5 'H' 5 'NHCl', garam asid conjugate, kation pyridinium, 'C' 5 'H' 5 'NH' (+). Example 4: (a) Calculate the pH of a 0.500 L buffer solution composed of 0.700 M formic acid (HCOOH, K a 1.77 x 10¯ 4) and 0.500 M sodium formate (HCOONa).(b) Calculate the pH after adding 50.0 mL of a 1.00 M NaOH solution. The Henderson Hasselbalch Equation is often used to calculate the pH of buffers and buffer systems. pH values are to be read on a scale of 1 to 14, with 1 being acidic and 14 being basic.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

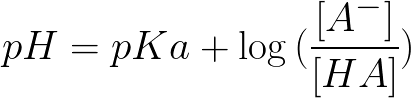
 RSS Feed
RSS Feed
